|
4/11/2024 0 Comments Dorsal column stimulator trial  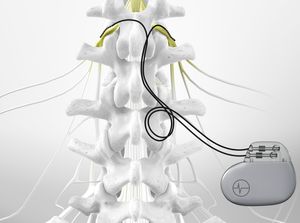
The methodology used to place the SCS leads was similar between all investigators, and this was verified during pre-study discussions. Description of SCS Lead Placement Technique Ten patients were entered sequentially by a physician who anchors his leads with suture and tape, and 10 subjects were entered sequentially by two physicians who use tape only for anchoring. Twenty patients age 18 or greater, male and female, who were selected for an SCS trial per routine clinical decision making in our pain practice, were entered sequentially into the study. Primary Aimsĭetermine how much movement (measured in millimeters superior or inferior) occurs in the location of percutaneous trial SCS leads between the time of insertion and the end of a 3-day trial (time of removal).ĭetermine if the method of anchoring affects the amount of lead movement.įollowing Institutional Review Board approval, consent was obtained for all subjects prior to study entry. 
The purpose of this study was to investigate whether percutaneous SCS leads migrate during the course of a 3-day trial, and whether the skin anchoring method used influenced the amount of migration. With this study we formally investigated lead migration in a prospective manner. Anecdotally, trial leads are often observed to move inferiorly within the epidural space by the third day of the trial. We currently obtain additional X-rays just prior to removal of the trial lead(s) to verify their location at the conclusion of the trial and this information has been incorporated in the decision-making process to help identify the best location for final paddle lead placement. In the past at our institution, the permanent paddle electrode was placed by the Neurosurgeon at the exact position where the trial lead(s) were located (as documented by the X-ray taken in the operating room) at the time of initial placement at the beginning of the trial. However, we have found good therapeutic success by meticulously identifying the location of optimal stimulation during the trial phase, and communicating this information effectively to the Neurosurgeon for planning the final surgical procedure. In our practice, during the surgical placement of the paddle electrode, we do not wake the patient in an attempt to try and verify stimulation coverage-we have found this problematic. It has been reported that paddle electrodes have less propensity for migration, utilize less energy, and may perhaps be more effective at relieving axial back pain. If a trial is successful (greater than 50% pain relief) we most often recommend placement of a permanent insulated surgical paddle electrode. In our current practice percutaneous SCS trial lead(s) are placed into the epidural space during outpatient surgery and remain in the epidural space for 3 days. The precise location for placement of the paddle electrode must be appropriately identified because a miscalculation can mean the difference between success and failure of the final implant. For patients who go on to have a surgical paddle electrode placed for the permanent stimulator, this latter point is of vital importance. The purpose of such a trial is twofold: to verify that SCS effectively relieves the patient's pain, and to identify the precise region of the spinal cord that produces this effect. Most commonly, percutaneous leads are placed for a trial (of variable duration), and are removed at trial completion. A recently published comprehensive report regarding the standard of care details many of the variations in methodology. There are a variety of generally accepted methods for performing a trial of spinal cord stimulation (SCS). Anchoring the trial electrodes to the skin with a suture and tape results in significantly greater inferior migration when compared with anchoring the lead with tape only. Percutaneous SCS trial electrodes migrate significantly, inferiorly, during a 3-day trial. A t-test revealed a significant difference between the groups ( t = 3.9, P= 0.001).Ĭonclusions. The average movement for leads anchored with tape only was 8.72 mm (SD = 5.77), inferiorly while movement for leads anchored with suture and tape was 24.49 mm (SD = 11.3), inferiorly. Using a standardized method, SCS leads were measured and movement was calculated. A standardized X-ray protocol of lead position was obtained immediately following lead placement and upon completion of the trial. Ten leads were anchored with suture and tape and 10 were anchored with tape only. Twenty patients were prospectively enrolled. Investigate whether percutaneous spinal cord stimulator (SCS) leads migrate significantly during a 3-day trial, and determine whether the skin anchoring method influences lead migration. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
